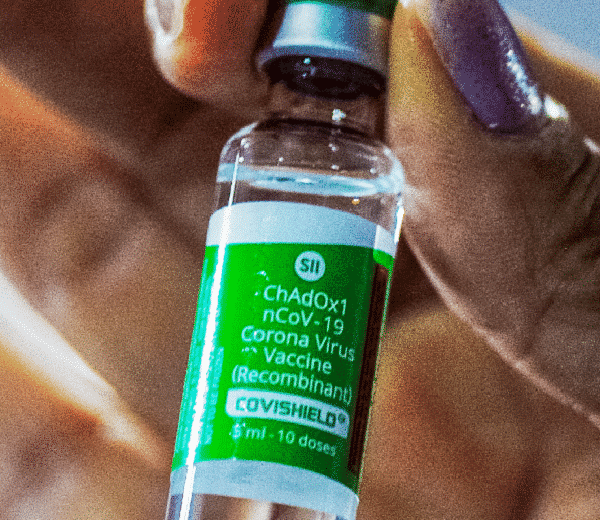
The Australian Government’s Therapeutic Goods Administration (TGA) has ruled that the Covishield (AstraZeneca/Serum Institute of India) vaccine is a “recognised” vaccine.
The announcement comes close on the heels of another announcement of resumption of travel to India for vaccinated individuals.
Both announcements have caused much relief – and jubilation – within Australia’s Indian community who are waiting to be reunited with family. After COVID wreaked havoc in India earlier this year, many Indian-Australians have waited patiently to be able to travel back to India to meet recovered and bereaved family members.
Amongst the international student community still stranded in India, this will no doubt see similar relief, although more announcements by the government for this sector are still awaited.
The murmurings in India regarding recognition of Covishield had turned into a roar in recent weeks, with Parliamentarian Shashi Tharoor even declining an invite to the UK in protest against its attitude to the Indian vaccine.
This is now a thing of the past.
The UK Medicines and Health products Regulatory Agency, as well as Health Canada, have now recognised Covishield – both are considered “Comparable Overseas Regulators” by the TGA. Italy has since granted similar approval.
In recognising Covishield, TGA noted, “Covishield (AstraZeneca/Serum Institute of India) is manufactured using the same ChAdOx1-S recombinant virus as the AstraZeneca (Vaxzevria) vaccine to produce the same dose of virus in the final product. The two are considered interchangeable by the World Health Organisation. TGA considers COVISHIELD to have the same clinical efficacy as Vaxzevria for this assessment.
Therefore, the clinical efficacy and effectiveness data for (AstraZeneca) are relevant in this case. The average Vaccine Efficacy and Effectiveness against symptomatic infection is 65% and severe infection and/or hospitalisation is 85%.”
It stated clearly that this information is “advice only and has no standing in law and does not represent assessment for regulatory approval within Australia. It will help inform decisions made elsewhere in Government to support incoming travel across Australia’s international borders in the coming months.”
It clarified though that “while certain vaccines may be considered by the TGA as “recognised” decisions on inbound travel are made by the Department of Home Affairs. State and Territory governments, or organisations such as universities, may apply additional considerations around vaccine requirements post-border.”
The advice comes after data was compared for two-dose schedules of selected vaccines not registered in Australia with that for vaccines currently in use here.